-
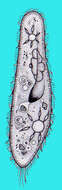
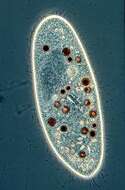
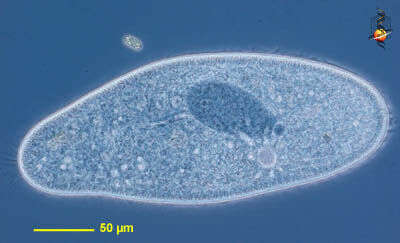
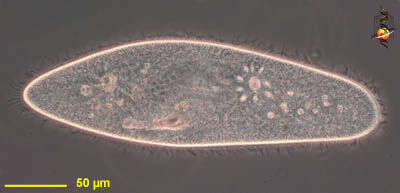
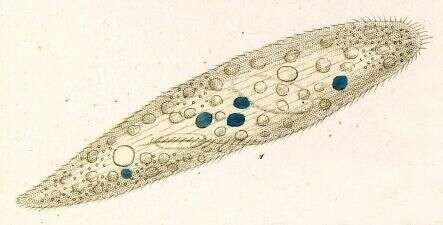
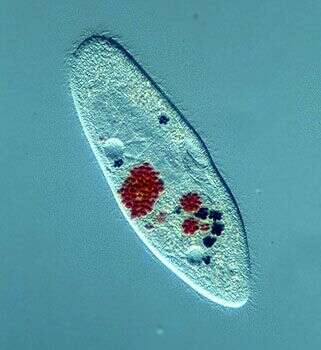
Paramecium (caudatum) (par-a-mee-see-um) is a very familiar genus of ciliates. They eat bacteria and have the mouth recessed in a buccal cavity, and the cell is often shaped with a scoop leading to the mouth. There are cilia all over the body with a caudal tuft of longer cilia at the back of the body. Usually with a layer of extrusomes (trichocysts) under the cell surface and a large oval macronucleus. Contractile vacuoles star-shaped. Most species are elongate, although this particular individual has been squashed so that we can see the nuclei. The (morpho)species are distinguished by morphology of the nuclei. This species is P. caudatum, which a single rather globular macronucleus lying alongside (4 o clock) the macronucleus. Phase contrast.
-
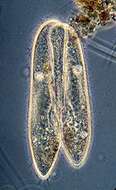
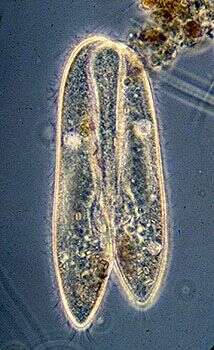
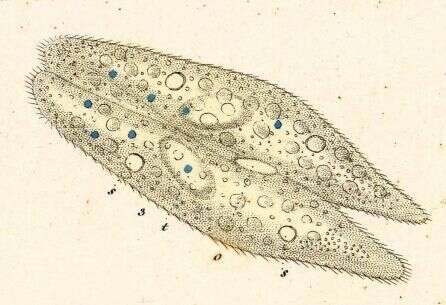
Paramecium (caudatum) (par-a-mee-see-um) is a very familiar genus of ciliates. They eat bacteria and have the mouth recessed in a buccal cavity, and the cell is often shaped with a scoop leading to the mouth. This cell has been compressed so that we can see various organelles. There are cilia all over the body with a caudal tuft of longer cilia at the back of the body. Usually with a layer of extrusomes (trichocysts) under the cell surface and a large oval macronucleus. Contractile vacuoles star-shaped. Phase contrast.
-
-

Detail of the oral apparatus and surrounding infraciliature (anterior ventral aspect) of the hymenostome ciliate Paramecium caudatum (Ehrenberg, 1833). The preoral and postoral sutures (lines of convergence of kinetal fields) can be clearly seen. The kinetids with their kinetodesmal fibers are clearly visible. The kinetodesmal fibers are periodically striated bundles of fibrils arising near the base of somatic kinetids (the posterior one if kinetids are paired) in ciliates. The kinetodesmal fibers extend anteriorly and to the right of their kinety (this is the Law of desmodexy). This provides a means of determining an anterior, posterior and right/left orientation in ciliates. Here the kinetodesmal fibers are longer than the interkinetal distance and therefore overlap giving the appearance of a longitudinal interkinetal line. The macronucleus and the single large micronucleus are seen anterior to the oral aperture. Silver carbonate stain (see Foissner, W.Europ. J. Protistol.27,313-330;1991). Specimen collected from freshwater pond near Boise, Idaho July 2004. Brightfield.
-
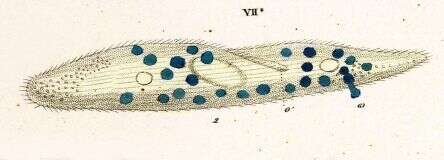
These two individuals have fused toigether and wille xchange haploid nuclei as part of a process of sexual activity. Ciloiates can divide asexually with mitotic division of the micronuclei and an a-mitoitic division of the macronucleus. Periodically, the cells go through a sexual process in which cells join at the mouth region, the macronucleus breaks down, micronuclei undergo meiosis, haploid nuclei are exchanged, they then fuse and the resulting product divides to create new micronuclei and macronuclei.
-
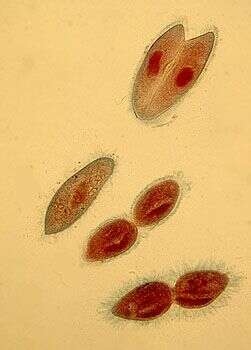
In this stained preparation, the nuclei are red. There is a single normal cell, a pair of cells joined in conjugation and two cells in the process of asexual division - which involves a division furrow cutting through the middle of the cells.
-
This single cell has been fixed and stained with a dye that shows up the nuclei. The image clearly shows the large macronucleus and the smaller, but still substantial. micronucleus. The arrangement of the nuclei is one of the criteria used to distinguish the species in this genus.
-
Passion. This image goes with the one of Eufolliculina. In this case this is a knitted contractile vacuole with the radiating canals, ampullae, vacuole and vacuolar pore. Knitted by June Hornby.
-
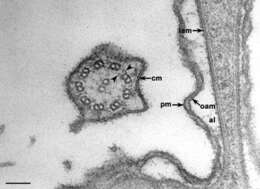
Higher magnification that shows the bilayer nature of the ciliary membrane (cm), plasma membrane (pm) and the outer (oam) and inner (iam) alveolar sac membranes of the alveoli (al). The 9-doublet + 2-singlet microtubular axonemal system of the cilium is seen in cross section (also see Figure 8). Two dynein arms (arrowheads) extend from each doublet. Cacodylate buffered fixative. EM taken on 9/27/68 by R. Allen with Philips 300 TEM. Neg. 60,400X. Bar = 0.1 micron.
This image is available in Richard Allen's collection.
-

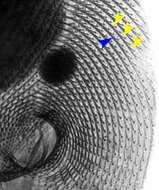
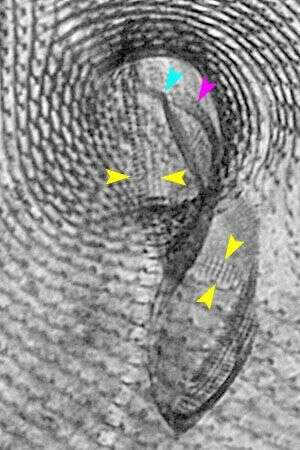
Detail of the oral apparatus and surrounding infraciliature (anterior ventral aspect) of the hymenostome ciliate Paramecium caudatum (Ehrenberg, 1833). The kinetodesmal fibers are periodically striated bundles of fibrils arising near the base of somatic kinetids (the posterior one if kinetids are paired) in ciliates. The kinetodesmal fibers extend anteriorly and to the right of their kinety (yellow arrowheads). This is the Rule of Desmodexy which states "true kinetoodesmata,when present, lie to or extend (anteriad) to the 9organism's) right of the kinety (line of kinetosomes) with which they are associated". This provides a means of determining an anterior, posterior and right/left orientation in ciliates. Here the kinetodesmal fibers are longer than the interkinetal distance and therefore overlap giving the appearance of a longitudinal interkinetal line (blue arrowhead). The macronucleus and the single large micronucleus are seen anterior to the oral aperture. Silver carbonate stain (see Foissner, W.Europ. J. Protistol.27,313-330;1991). Specimen collected from freshwater pond near Boise, Idaho July 2004. Brightfield.
-
Detail of the oral apparatus and surrounding infraciliature (anterior ventral aspect) of the hymenostome ciliate Paramecium caudatum (Ehrenberg, 1833).between the two sets of yellow arrowheads are the four loosely spaced kineties of the "quadrulus" (M3). The pink and light blue arrowheads mark the first (M1) and second (M2) adoral membranelles.Silver carbonate stain (see Foissner, W.Europ. J. Protistol.27,313-330;1991). Specimen collected from freshwater pond near Boise, Idaho July 2004. Brightfield.
-
-
-
-
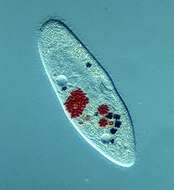
Paramecium fed with dead bacteria stained red and with some indian ink added, such that food vacuoles form that are red and black in colour. These are normal sized food vacuoles (compare with the paramecia that have been fed with yeast cells). The mouth cilia form the dark band located near the middle of the cell. The macronucleus is anterior, a contractile vacuole with radiating collecting canals and expanded ampullae is evident in the back of the cell. The posterior end of the cell has a tuft of caudal cilia that are longer than the cilia over the rest of the body.
-
This cell has been allowed to feed on yeast cells that have been stained with neutral red. This is a simple way to demonstrate the ingestion of the particles, and the changes that occur within the food vacuole as it is processed within the ciliate cell. The stain changes to a blue colour as the contents of the vacuole are acidified. Yeast cells are larger than the normal food of Paramecium (they mostly eat bacteria). The food vacuoles that form can be aberrant (in this case one is very large) and the behaviour of the food vacuoles inside the cell is also disturbed. The demonstration of feeding by Paramecium is best done with stained bacteria.
-
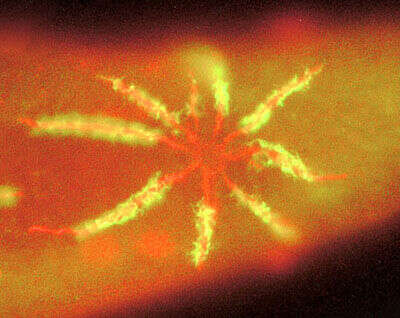
Immunofluorescence image showing the distribution of two antibodies. The green comes from the fluorescein die and stains an ATPase enzyme that is associated with the the spongiome; while the red antibody (Texas red) labels a protein that is associated with the radiating collecting canals, the ampullae the pore - and also the cell surface.
This image is available in Richard Allen's collection.
-
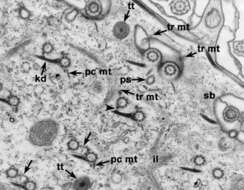
Tangential section through the cell surface shows the pairs of basal bodies in cross section. The ends of kinetodesmal fibers (kd) point anteriorly in the cell, the postciliary ribbons (pc mt) arise from the posterior right side of the posterior basal body of a pair and transverse ribbons (tr mt) arise from the left side of both basal bodies of a pair. The more anterior basal body of a pair is linked to the posterior basal body by a striated fiber (arrows) and to the adjacent kinetodesmal fiber by a short filamentous connection (arrowhead). ps, parasomal sacs; il, infraciliary lattice; sb, striated band; tt, trichocyst tip. EM taken on 1/12/73 by R. Allen with Hitachi HU11A TEM. Neg. 14,900X. Bar = 0.5 microns.
This image is available in Richard Allen's collection.
-
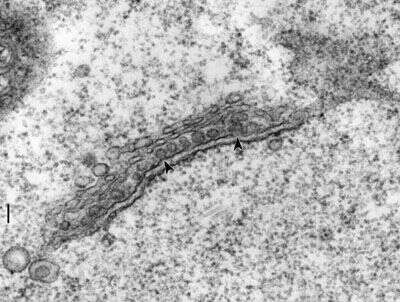
Dictyosome (Golgi apparatus) showing that ribosomes are attached to the cytosolic side of the transition zone of the ER. COP-II coats presumably allow the ER membrane to accumulate protein cargo and to evaginate (arrowheads) into a pit which will pinch off into transition vesicles. These then fuse with the Golgi stack which in paramecia consists of only two or three cisternae. Glycosylation of the proteins may occur in the cisternae which may target the proteins to lysosomes, to the plasma membrane, or to the endosomal system. Paramecium does not have sialic acid as the terminal sugar of its complex carbohydrate as part of its glycosylated proteins and lipids. Bar = 0.1 microns.
This image is available in Richard Allen's collection.
-
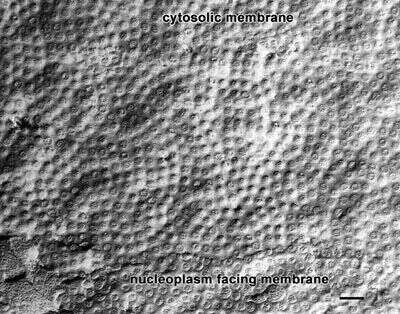
A freeze-fractured view of the nuclear envelope. The fracture in the middle of the figure exposes the cytosolic facing membrane. This can be distinguished by a fractured cytoplasmic organelle at the top of the picture. The lower part of the picture shows the nucleoplasm-facing membrane of the envelope. The nuclear pores penetrate through both membranes. It is not clear if the membranes have actually fractured along their hydrophobic centers or if the true surfaces of these two membranes are being viewed. EM taken on 2/16/76 by R. Allen with Hitachi HU11A TEM. Neg. 14,579X. Bar = 0.25 micron.
This image is available in Richard Allen's collection.
-
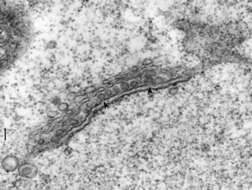
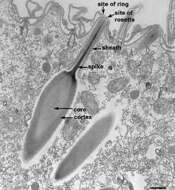
The body of the trichocyst is composed of a core and a cortex which have slightly different electron diffracting properties, thus the cortex is darker since it scatters more electrons. An electron transparent zone surrounds the cortex and separates the trichocyst membrane from the body. The trichocyst tip is composed of a central spike covered by a sheath. The spike is nail-shaped with its head resting on the trichocyst body. The trichocyst membrane completely surrounds both trichocyst body and sheath. (Details of trichocyst fine structure were recorded by Bannister, J. Cell Sci. 11:899-929, 1972.) EM taken on 4/6/78 by R. Allen with Hitachi HU11A TEM. Neg. 14,750X. Bar = 0.5 micron.
This image is available in Richard Allen's collection.
-
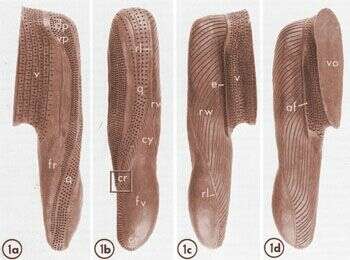
A clay model of the space occupied by the buccal cavity, its connection to the vestibulum (v) and the nascent food vacuole (fv) arising from the cytopharynx (cr). Dots indicate sites of basal body/cilium complexes of the somatic ciliature on the vestibulum and the membranelles of the ventral peniculus (vp), the dorsal peniculus (dp) and the quadrulus (q). In addition the endoral membranelle (e) forms a single row along the right side of the vestibulum/buccal cavity border. A field of unorganized basal bodies, the anarchic field (af), lies next to the endoral membranelle. An enlargement of the box labeled cr is presented in Figure 28. fr, filamentous reticulum; rl, groove in right lip of cytopharynx; rw, ribbed wall; cy, cytostome-cytopharynx complex; vo, vestibular opening. Published in J. Cell Biol. 63:904-922, 1974.
This image is available in Richard Allen's collection.
-
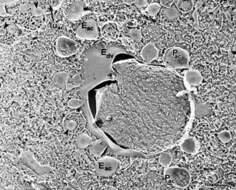
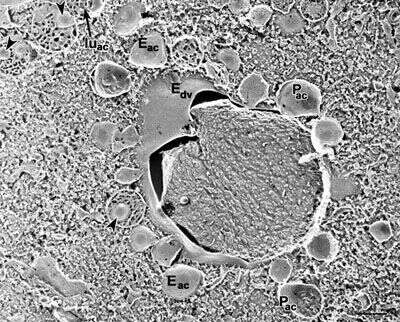
Quick-freeze deep-etch images show a more three dimensional view of acidosomes surrounding a phagosome. Acidosomes are unique in that they contain indentations of their membrane (arrowheads). The lumen (luac) has a honeycombed appearance, also unique to these vesicles. Their E-face (Eac) is almost free of IMPs while their P-face (Pac) has many large particles which leave pits in the E-face. Acidosomes maintain a close association with the phagosome membrane. Edv, E-face of phagosome. EM taken on 5/26/92 by R. Allen with Zeiss 10A TEM. Neg. 9,780X. Bar = 0.5 microns.
This image is available in Richard Allen's collection.
-
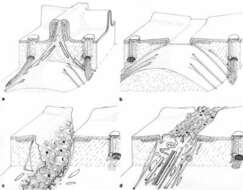
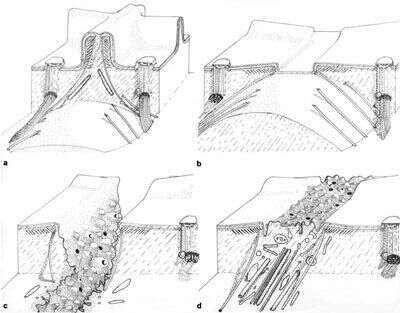
Drawing of the cytoproct illustrating four views as it changes shape during vacuole defecation. a) A view of the short segment of the closed and inactive cytoproct. Single microtubules extend into the cytosol from the top of the ridge which is distinguished from other ridges by piles of unique fibers. Bundles of microtubules also extend inward from the adjoining basal bodies. b) The microtubules contact the spent vacuole and move the vacuole toward the cellâs surface. Ultimately the microtubules pull the vacuole and ridge apex together where fusion between the vacuole and plasma membrane can occur. c) Following fusion the spent vacuole membrane tubulates and fission results in release of this membrane back into the cell. Tubulation is promoted by actin filaments. d) The ridge will reform when all spent vacuole membrane is retrieved. Published in J. Cell Sci. 14:611-631, 1974.
This image is available in Richard Allen's collection.